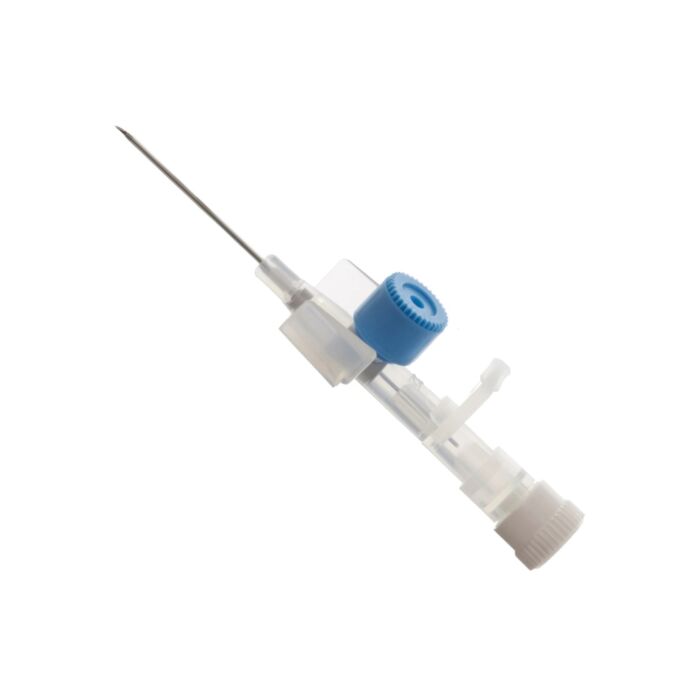
BD Venflon™ Peripheral IV Catheter | 22G x 25mm | Single Unit
The BD Venflon™ Peripheral IV Catheter is designed for sterile, single-use and features an integrated injection port and valve for efficient peripheral catheterisation. This blue, 22 G x 25 mm catheter facilitates the needle-free administration of bolus medications during infusion, enhancing patient comfort and safety.
Key Features:
Integrated Injection Port and Valve: Allows for the direct administration of medications without the need for needle access, maintaining system integrity.
Enhanced Insertion Design: The back-cut needle bevel and tapered end of the PTFE catheter ensure smoother insertion and reduced risk of vein damage, promoting patient comfort.
Hygienic Wing Position: Strategically placed wings decrease the risk of bacterial contamination by avoiding direct contact between the catheter hub's rear and the patient's skin. These wings also aid in securing the catheter when used with BD Veca-C™ and BD Veca-Fix™ IV dressings.
BD Luer-Lok™ Compatibility: Ensures secure and reliable connections with other Luer-Lok™ system components.
Single-Use Sterility: Guaranteed sterile usage to prevent infection and cross-contamination in clinical settings.
| Size | |
|---|---|
| Size |
| Absorbency | |
|---|---|
| Accuracy | |
| Application | |
| Arch | |
| Automation | |
| Battery Life | |
| Bristles | |
| Capacity | |
| Cleaner Type | |
| Closure Type | |
| Colour | Blue |
| Colour Temperature | |
| Configuration | |
| Container | |
| Cuff | |
| Density | |
| Depth | |
| Display | |
| Doors | |
| Door Type | |
| Speed | |
| Filter Level | |
| Fitment | |
| Fold | |
| Fragrance | |
| Further Information | |
| Gauge | 22 |
| Gender | |
| Grade | |
| Grit | |
| gsm | |
| Height | |
| Housing Construction | |
| Illumination | |
| Impervious | |
| Instrument Size | |
| Insufflation Bulb | |
| Interface | |
| Length | |
| Light Intensity | |
| Magnification | |
| Magnification/Working Distance | |
| Material | |
| Maximum Temperature | |
| Max Height | |
| Mhz | |
| Minimum Height | |
| Minimum Temperature | |
| Needle Diameter | |
| Needle Length | 25mm |
| Oxygen Percent | |
| Pack Size | 1 |
| Panels | |
| Ply | |
| Polorisation | |
| Powder | |
| Power Source | |
| Precision | |
| Prefilled | |
| Reinforced | |
| Resistance | |
| Ruler | |
| Shape | |
| Shelves | |
| Side | |
| Size | |
| Size | |
| Softness | |
| Spot Size | |
| Sterility | |
| Strength | |
| Thickness | |
| Tip | |
| Trays | |
| Tube Length | |
| Tube Type | |
| Type | |
| Type of Light | |
| Usage | |
| Viewing window | |
| Volume | |
| Waste Type | |
| Width | |
| Working Distance |
| GTIN | |
|---|---|
| Manufacturer | Becton Dickinson |
| MPN | BV391451 |
| Warranty |
| Delivery Time |
|---|
The BD Venflon™ Peripheral IV Catheter is designed for sterile, single-use and features an integrated injection port and valve for efficient peripheral catheterisation. This blue, 22 G x 25 mm catheter facilitates the needle-free administration of bolus medications during infusion, enhancing patient comfort and safety.
Key Features:
Integrated Injection Port and Valve: Allows for the direct administration of medications without the need for needle access, maintaining system integrity.
Enhanced Insertion Design: The back-cut needle bevel and tapered end of the PTFE catheter ensure smoother insertion and reduced risk of vein damage, promoting patient comfort.
Hygienic Wing Position: Strategically placed wings decrease the risk of bacterial contamination by avoiding direct contact between the catheter hub's rear and the patient's skin. These wings also aid in securing the catheter when used with BD Veca-C™ and BD Veca-Fix™ IV dressings.
BD Luer-Lok™ Compatibility: Ensures secure and reliable connections with other Luer-Lok™ system components.
Single-Use Sterility: Guaranteed sterile usage to prevent infection and cross-contamination in clinical settings.
| Size | |
|---|---|
| Size |
| Absorbency | |
|---|---|
| Accuracy | |
| Application | |
| Arch | |
| Automation | |
| Battery Life | |
| Bristles | |
| Capacity | |
| Cleaner Type | |
| Closure Type | |
| Colour | Blue |
| Colour Temperature | |
| Configuration | |
| Container | |
| Cuff | |
| Density | |
| Depth | |
| Display | |
| Doors | |
| Door Type | |
| Speed | |
| Filter Level | |
| Fitment | |
| Fold | |
| Fragrance | |
| Further Information | |
| Gauge | 22 |
| Gender | |
| Grade | |
| Grit | |
| gsm | |
| Height | |
| Housing Construction | |
| Illumination | |
| Impervious | |
| Instrument Size | |
| Insufflation Bulb | |
| Interface | |
| Length | |
| Light Intensity | |
| Magnification | |
| Magnification/Working Distance | |
| Material | |
| Maximum Temperature | |
| Max Height | |
| Mhz | |
| Minimum Height | |
| Minimum Temperature | |
| Needle Diameter | |
| Needle Length | 25mm |
| Oxygen Percent | |
| Pack Size | 1 |
| Panels | |
| Ply | |
| Polorisation | |
| Powder | |
| Power Source | |
| Precision | |
| Prefilled | |
| Reinforced | |
| Resistance | |
| Ruler | |
| Shape | |
| Shelves | |
| Side | |
| Size | |
| Size | |
| Softness | |
| Spot Size | |
| Sterility | |
| Strength | |
| Thickness | |
| Tip | |
| Trays | |
| Tube Length | |
| Tube Type | |
| Type | |
| Type of Light | |
| Usage | |
| Viewing window | |
| Volume | |
| Waste Type | |
| Width | |
| Working Distance |
| GTIN | |
|---|---|
| Manufacturer | Becton Dickinson |
| MPN | BV391451 |
| Warranty |
| Delivery Time |
|---|
The BD Venflon™ Peripheral IV Catheter is designed for sterile, single-use and features an integrated injection port and valve for efficient peripheral catheterisation. This blue, 22 G x 25 mm catheter facilitates the needle-free administration of bolus medications during infusion, enhancing patient comfort and safety.
Key Features:
Integrated Injection Port and Valve: Allows for the direct administration of medications without the need for needle access, maintaining system integrity.
Enhanced Insertion Design: The back-cut needle bevel and tapered end of the PTFE catheter ensure smoother insertion and reduced risk of vein damage, promoting patient comfort.
Hygienic Wing Position: Strategically placed wings decrease the risk of bacterial contamination by avoiding direct contact between the catheter hub's rear and the patient's skin. These wings also aid in securing the catheter when used with BD Veca-C™ and BD Veca-Fix™ IV dressings.
BD Luer-Lok™ Compatibility: Ensures secure and reliable connections with other Luer-Lok™ system components.
Single-Use Sterility: Guaranteed sterile usage to prevent infection and cross-contamination in clinical settings.